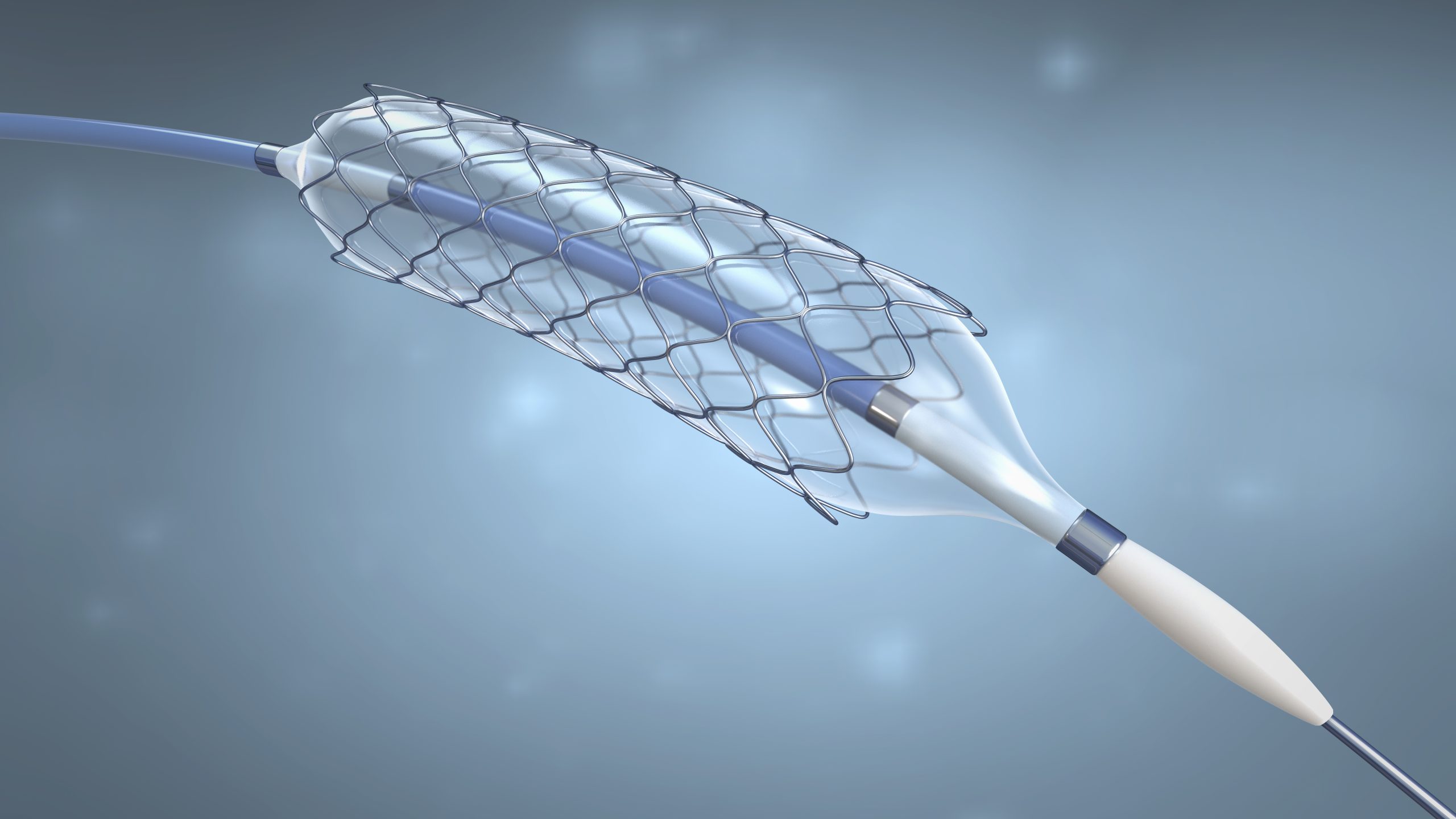
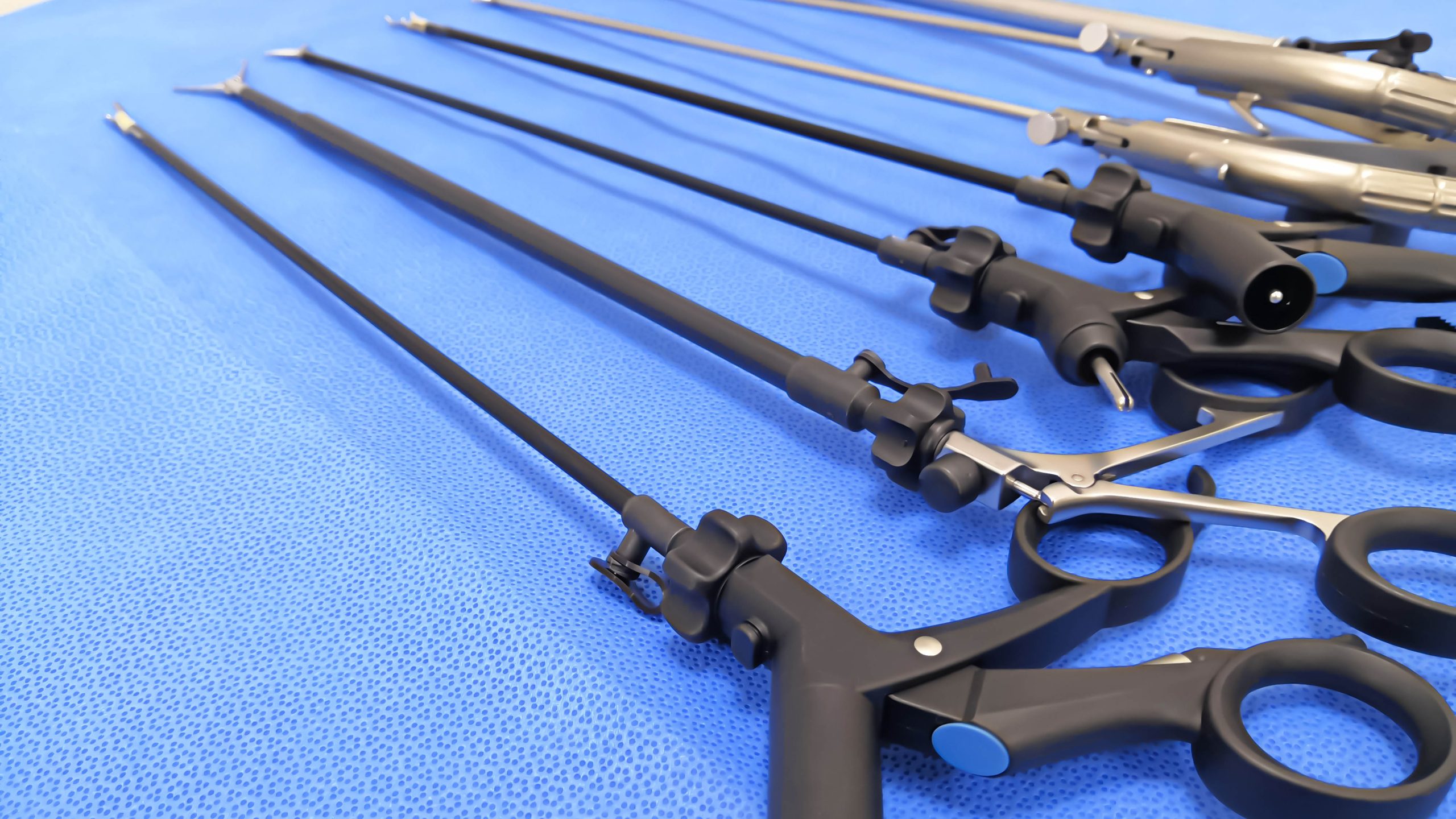
Specialty materials for minimally invasive procedures
Explore a comprehensive portfolio of advanced polymers and specialty materials selected for minimally invasive devices across surgical, diagnostic, and therapeutic applications.
Explore a comprehensive portfolio of advanced polymers and specialty materials selected for minimally invasive devices across surgical, diagnostic, and therapeutic applications.

Minimally invasive medical devices place exceptional demands on material performance. As components become smaller, more complex, and more tightly integrated into clinical workflows, material choice directly influences device precision, durability, and ease of processing. Polymers and composites used in these applications must support tight tolerances, repeated sterilization, chemical exposure, all while exhibiting increasingly sophisticated mechanical and optical functions.
Mitsubishi Chemical Group partners with manufacturers of minimally invasive medical devices to address these challenges through a broad portfolio of specialty products and deep experience in material science for minimally invasive procedures. From early material selection through validation and scale-up, we help you optimize performance, reliability, and regulatory readiness across a wide range of minimally invasive applications.
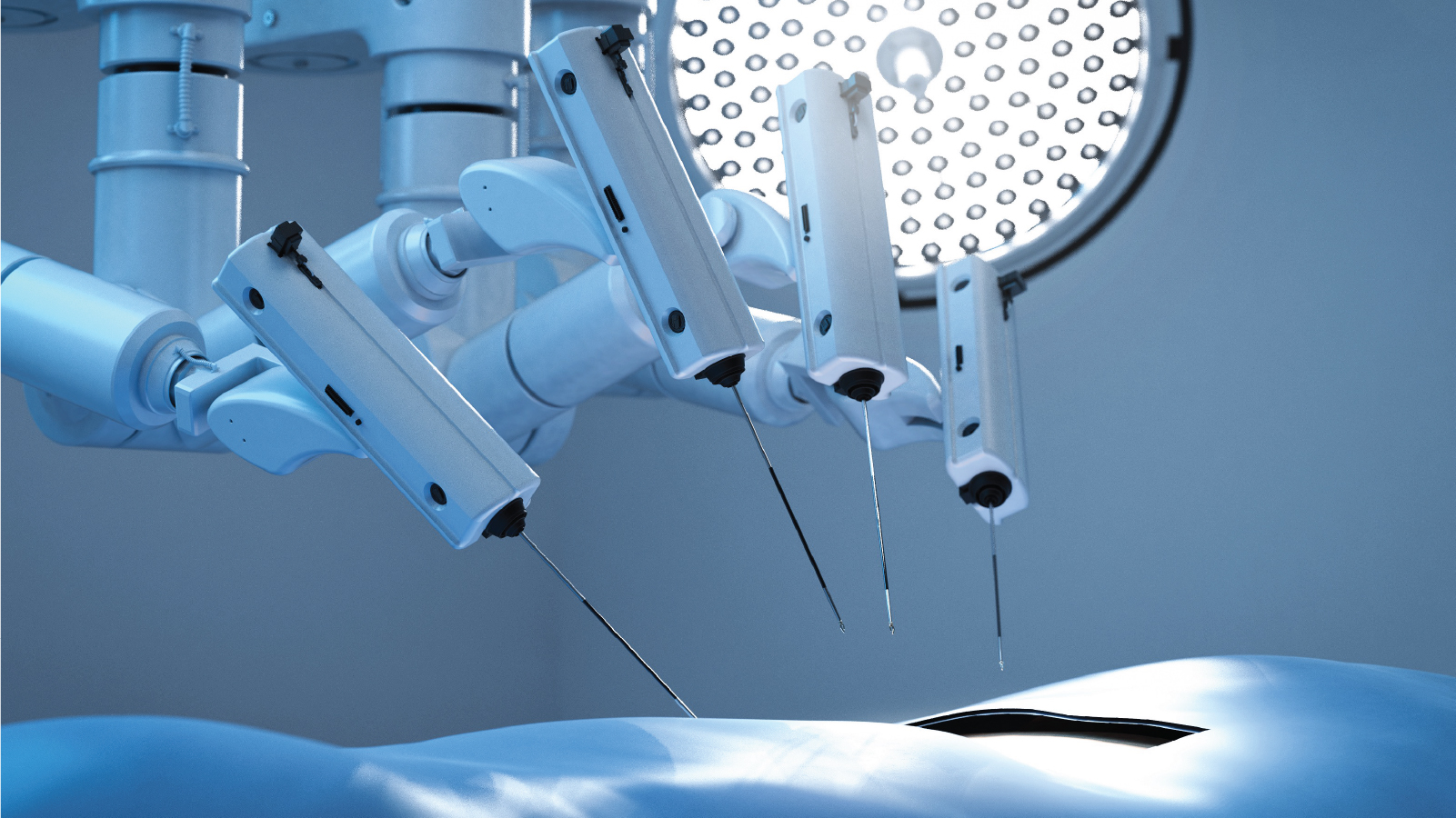
The effectiveness and efficiency of robotic systems in healthcare rely in no small part on material performance. Thermoplastic materials in medical robotic applications must deliver exceptional mechanical precision, long-term reliability, and dimensional stability under repeated sterilization. Critical components such as articulated instruments, housings, and actuation elements must maintain tight tolerances across autoclave, plasma, or chemical sterilization cycles while supporting smooth motion, low friction, and consistent force transmission.
Selecting materials for robotic applications presents complex trade-offs between stiffness and fatigue resistance, weight reduction, radiolucency, and compatibility with advanced imaging environments. As systems become more compact and multifunctional, thermoplastics and composites must also enable intricate geometries and integrate seamlessly with metal or electronic components. Furthermore, selected materials must also support the robotic systems in meeting stringent regulatory requirements.
Mitsubishi Chemical Group partners with developers of robotic systems from early design and prototyping through scalable manufacturing. By tailoring high-performance thermoplastic materials to specific mechanical, thermal, and sterilization demands, we help you engineer robotic platforms that deliver enhanced accuracy and durability throughout the device lifecycle.
Select materials
Intravenous delivery systems depend on materials that exhibit consistent performance across a wide range of therapies, flow conditions, and clinical environments. Catheters, hubs, connectors, and tubing must combine chemical resistance, dimensional stability, and flexibility, and they must maintain these performance characteristics through sterilization, storage, and use.
Material selection for IV systems is increasingly complex as formulations expand to include biologics, lipid-based drugs, and cytotoxic compounds. OEMs must balance considerations about kink resistance, insertion performance, and patient comfort with needs for low extractables, compatibility with sterilization methods, and evolving regulatory expectations. These performance factors are further compounded by processing requirements including reliable bonding, overmolding, and high-volume manufacturing.
Mitsubishi Chemical Group supports IV system development by helping you identify and tailor polymers optimized for mechanical performance, chemical compatibility, and processability. Through material expertise and application-specific testing, we help accelerate validation, support regulatory evaluation, and enable scalable production of next-generation intravenous devices.
Select materials
Use in minimally invasive procedures places high demands on the material performance of instruments and accessories. Components such as trocars, graspers, clip appliers, irrigation elements, and visualization accessories require polymers that balance mechanical strength, wear resistance, and dimensional stability with precise tolerances and ergonomic design. At the same time, the materials must withstand repeated clinical use and sterilization.
OEMs developing such devices face critical material decisions around reuse versus single-use, resistance to aggressive cleaning agents, and long-term durability across sterilization cycles. Optical clarity, surface finish, and friction properties can be equally important, particularly for visualization and access components where performance directly impacts procedural efficiency.
Mitsubishi Chemical Group works with medical device manufacturers to tailor advanced polymers for these demanding applications, accounting for mechanical loads, sterilization exposure, and manufacturing processes such as injection molding and overmolding. By aligning material properties with device design and lifecycle requirements, we help you engineer instruments and accessories that perform consistently over the entire product lifecycle.
Select materials