ChronoFlex™ S — the sweet spot
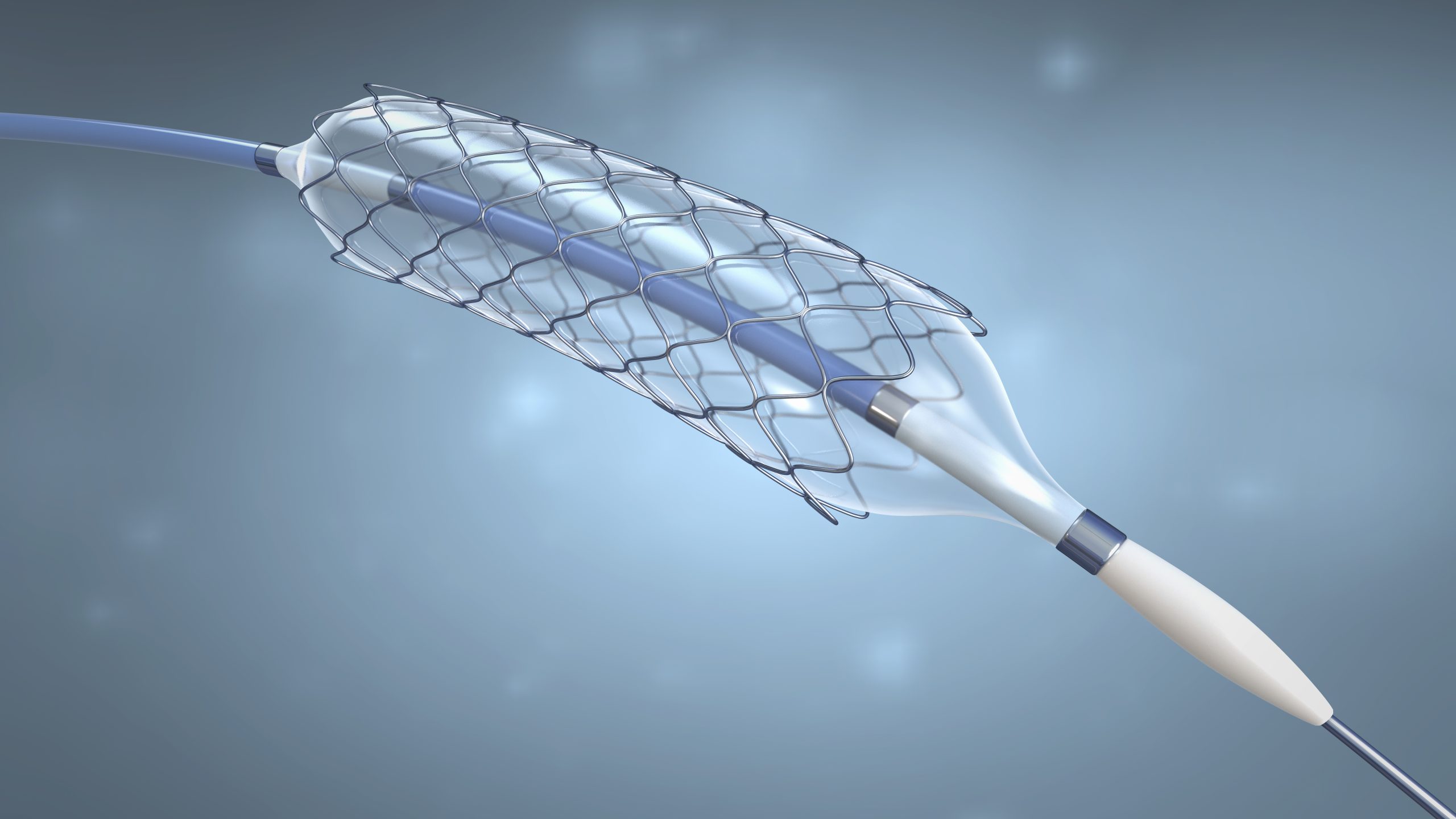
The ChronoFlex portfolio of products is known for its reliability and versatility for medical device applications. However, current products, including ChronoFlex C and ChronoFlex AL, have a hardness range of about 75A – 75D. To soften the product without compromising tensile strength or durability, our scientists used advanced polymer chemistry to develop a softer product without added plasticizers. The result: ChronoFlex S, a grade 60A TPU that delivers the softness and comfort previously only available with silicone while maintaining the superior strength, durability, and long-term stability of polyurethane – avoiding the conventional degradation risks of plasticized materials.
The balance of strength and softness may make ChronoFlex S suitable for more complex devices. “A neurovascular catheter needs to travel from the groin to the brain,” said Tokura. “The tip must be compliant to avoid damage to blood vessels, but the back end needs to be strong enough for the surgeon to push the catheter to the proper location. Combined, physicians have a device that can access sites in the body that wouldn’t be possible with a harder material.”
One of the most distinctive features of ChronoFlex S is its softening at in-body temperatures. Its unique polymer backbone responds to body temperature by becoming more conformable, potentially dropping from 60A to approximately 54A.
The formulation of ChronoFlex S does not utilize added plasticizers which may enhance long-term stability. Plasticizers can leach out over time, causing the material to harden and potentially fail. ChronoFlex may mitigate this risk by design. As a result, blooming/elution issues resulting from plasticizer-containing products are not expected with ChronoFlex S.
By integrating ChronoFlex S into medical products, medical device companies can potentially shorten development cycles and reduce processing complexity. ChronoFlex S is available in pellet, granule, or solution form and is compatible with extrusion, injection molding, compression molding, solution casting, dip molding/coating, and electrospinning—providing manufacturers with exceptional processing flexibility.
It can bond directly to other materials such as polyamide-type elastomers without adhesives or tie-layers, simplifying device construction. Aromatic TPU’s like ChronoFlex S are typically sterilized via ethylene oxide. Radiation-based sterilization methods may also be compatible depending on the specific device design and acceptable color change parameters, though validation is required for each application.
“The versatility of this TPU is excellent for applications requiring a soft material,” said Tokura. “You can process it by heat, dip-mold or cast film from solution. As a TPU without plasticizers, this product maintains processing method advantages while minimizing the potential for material degradation over time.”